
JMi ONConsulting
Providing additional keys and solutions to support your pharmaceutical drug research and early clinical development programs.
ABOUT
Michel JANICOT, Ph.D.
Founder & Managing Director, JMi ONConsulting

Ph.D. Biochemistry
Molecular and Cellular Biology
April 1988: Magna cum laude from Université Paris VII (France)

Postdoctoral Fellow
Biological Chemistry
1988 – 1992: The Johns Hopkins University (Baltimore, MD, USA)

30+ years hands-on experience in pharmaceutical drug research and early clinical development
350+ invited lectures, patents, book chapters, peer-reviewed publications, and abstracts
Rhône-Poulenc Rorer (Sanofi)
1992 – 1998: Vitry s/Seine (France)
Janssen Pharmaceutica (Johnson & Johnson)
1999 – 2012: Beerse (Belgium)
JMi ONConsulting
from 2012: Brussels (Belgium)
aPODD Foundation
from 2012: London (UK)
CURRENT ROLES

Founder and Managing Director
To provide additional keys and solutions to support your pharmaceutical drug research and early clinical development programs.
![]() Strategic preclinical project portfolio management
Strategic preclinical project portfolio management
![]() Drug Discovery
Drug Discovery
![]() Lead Optimization
Lead Optimization
![]() Medicinal Chemistry & Manufacturing (via network)
Medicinal Chemistry & Manufacturing (via network)
![]() Preclinical Development
Preclinical Development
![]() IND/CTA filing Process
IND/CTA filing Process
![]() Translational Medicine
Translational Medicine
![]() Early Clinical Development
Early Clinical Development
![]() Scientific Licensing
Scientific Licensing
![]() Fund raising campaigns (road show)
Fund raising campaigns (road show)

Co-Founder and Director/Trustee
“To accelerate the development and approval of new drugs to treat children with cancer. Specific drugs are not being developed and children gain only very limited benefit from the newer anti-cancer therapies that are approved for adults.”
aPODD Foundation is a registered charity in England and Wales and is a company limited by guarantee in England and Wales.
International scientific and technical expertise, and support.
20+ New Molecular Entities brought into first-in-human studies and further early clinical development.
CURRENT (PART-TIME) ASSIGNMENTS

Chief Development Officer
Ymmunobio
Zurich, Switzerland

Preclinical Development Advisor
Flamingo Therapeutics
Leuven, Belgium

Chief Development Officer
Onena Medicines
San Sebastian, Spain

Co-Founder & Board of Directors Member
aPODD Foundation
London, UK

Board of Directors Member
Experimentica
Kuopio, Finland
Preclinical/Phase 1 Development Advisor
Acctena AB
Åkers Styckebruk, Sweden
Scope & Capacities
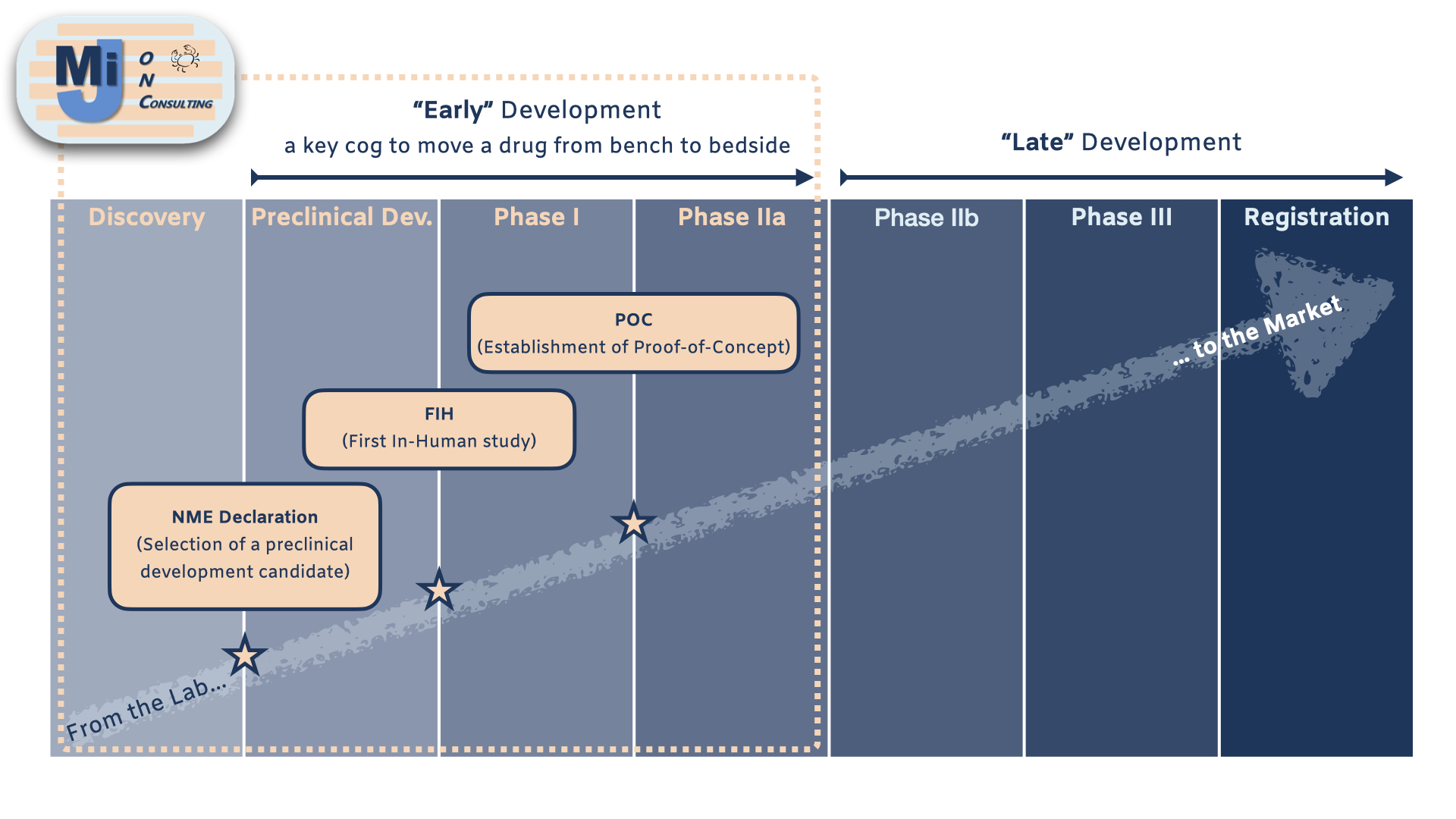

In-Licensing Opportunities
- Scouting and initial screening of potential opportunities to fit and complement your internal project pipeline
- Initial scientific evaluation and coordination of due diligence
Out-Licensing Opportunities
- Scouting and initial screening of potential partners
- Preparation of scientific information package for due diligence
- Preparation (coaching) of scientific presentations to potential partners
- Coordination and management of due diligence process
Internal Pharmaceutical Drug R&D Projects
- ‘C’ level management position
- Pharmaceutical Res. & early Dev. process, procedures and (regulatory/scientific) requirements
- Internal project evaluation (value proposition), management and scientific development
- Strategic and scientific project portfolio management
- Introduction to Contract Research Organizations (CROs) and outsourced activities : challenges & opportunities
- Coaching/preparation to (internal/external) scientific and strategic presentations
- Fund raising process
CONTACT

Michel Janicot
Founder & Managing Director


